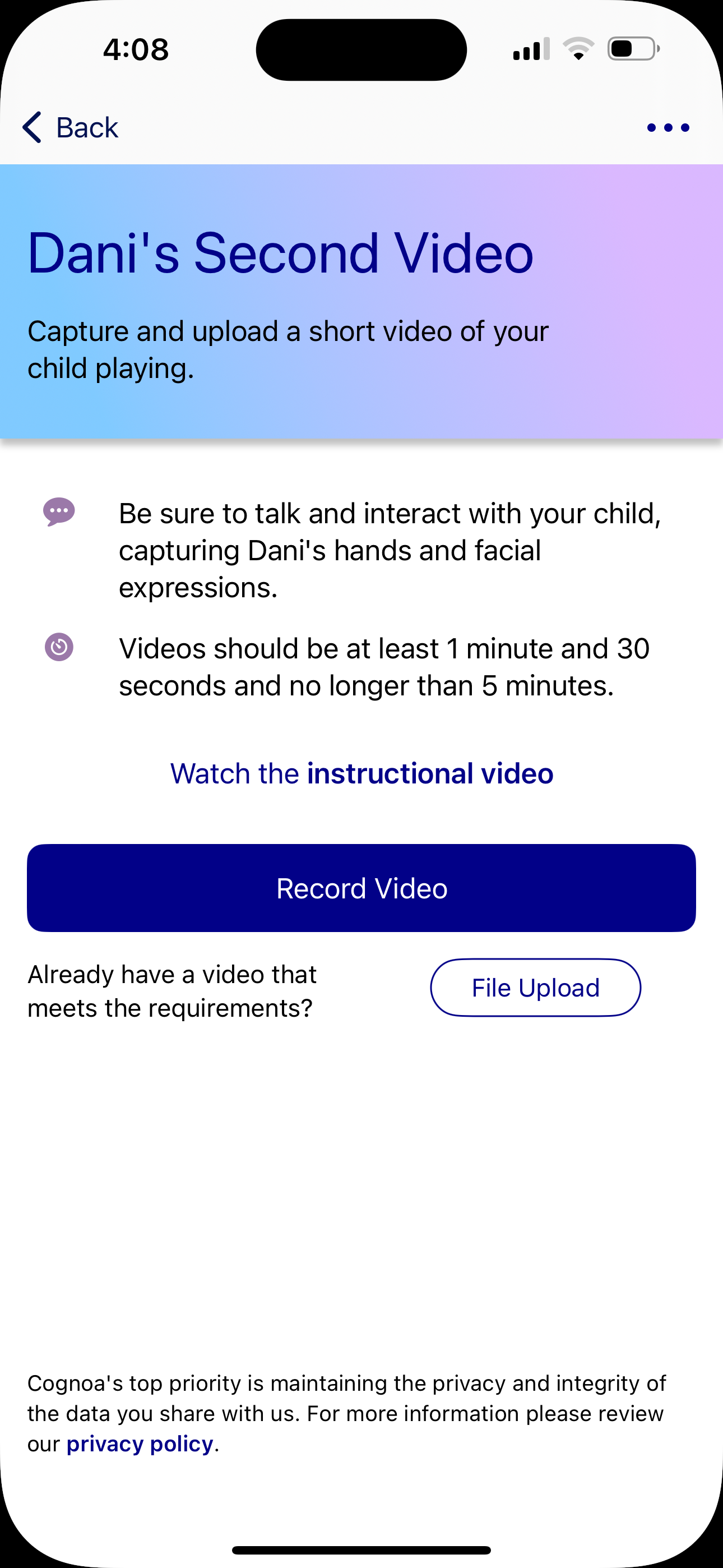
Beschreibung
Canvas Dx is for prescription use only. To find a prescriber go to cognoa.com or email [email protected].
Canvas Dx is the first and only FDA-authorized Software as a Medical Device (SaMD) that aids physicians in diagnosing autism spectrum disorder (ASD) in young children. Canvas Dx harnesses clinically validated artificial intelligence (AI) technology to aid physicians in diagnosing ASD in children between the ages 18-72 months who are at risk of developmental delay.
Canvas Dx incorporates 3 separate, user-friendly inputs:
1. A parent/caregiver questionnaire that asks about the child’s behavior and development collected via a parent/caregiver-facing app
2. A questionnaire completed by video analysts who review two videos of the child recorded by the parents/caregivers
3. An HCP questionnaire completed by a physician who meets with the child and a parent/caregiver, collected via a healthcare provider portal
The Canvas Dx algorithm evaluates all 3 inputs, generating a device output for the prescribing physician to utilize in combination with their clinical judgment.
Canvas Dx is not intended for use as a stand-alone diagnostic device but as an adjunct to the diagnostic process.
CONTACT:
Email: [email protected]
Indications for Use
Canvas Dx is intended for use by healthcare providers as an aid in the diagnosis of autism spectrum disorder (ASD) for patients ages 18 months through 72 months who are at risk for developmental delay based on concerns of a parent, caregiver, or healthcare provider.
The device is not intended for use as a stand-alone diagnostic device but as an adjunct to the diagnostic process. The device is for prescription use only (Rx only).
Contraindications
There are no contraindications to using Canvas Dx.
Precautions, Warnings
The Device is intended for use by healthcare professionals trained and qualified to interpret the results of a behavioral assessment examination and to diagnose ASD.
The Device is intended for use in conjunction with patient history, clinical observations, and other clinical evidence the HCP determines are necessary before making clinical decisions. For instance, additional standardized testing may be sought to confirm the Device output, especially when the Device result is not Positive or Negative for ASD.
Canvas Dx is intended for patients with caregivers who have functional English capability (8th grade reading level or above) and have access to a compatible smartphone with an internet connection in the home environment.
The Device may give unreliable results if used in patients with other conditions that would have excluded them from the clinical study.
Among those conditions are the following:
• Suspected auditory or visual hallucinations or with prior diagnosis of childhood onset schizophrenia
• Known deafness or blindness
• Known physical impairment affecting their ability to use their hands
• Major dysmorphic features or prenatal exposure to teratogens such as fetal alcohol syndrome
• History or diagnosis of genetic conditions (such as Rett syndrome or Fragile X)
• Microcephaly
• History or prior diagnosis of epilepsy or seizures
• History of or suspected neglect
• History of brain defect injury or insult requiring interventions such as surgery or chronic medication
The Device evaluation should be completed within 60 days of the time it is prescribed because neurodevelopmental milestones change rapidly in the indicated age group.
Ausblenden
Mehr anzeigen...
Canvas Dx is the first and only FDA-authorized Software as a Medical Device (SaMD) that aids physicians in diagnosing autism spectrum disorder (ASD) in young children. Canvas Dx harnesses clinically validated artificial intelligence (AI) technology to aid physicians in diagnosing ASD in children between the ages 18-72 months who are at risk of developmental delay.
Canvas Dx incorporates 3 separate, user-friendly inputs:
1. A parent/caregiver questionnaire that asks about the child’s behavior and development collected via a parent/caregiver-facing app
2. A questionnaire completed by video analysts who review two videos of the child recorded by the parents/caregivers
3. An HCP questionnaire completed by a physician who meets with the child and a parent/caregiver, collected via a healthcare provider portal
The Canvas Dx algorithm evaluates all 3 inputs, generating a device output for the prescribing physician to utilize in combination with their clinical judgment.
Canvas Dx is not intended for use as a stand-alone diagnostic device but as an adjunct to the diagnostic process.
CONTACT:
Email: [email protected]
Indications for Use
Canvas Dx is intended for use by healthcare providers as an aid in the diagnosis of autism spectrum disorder (ASD) for patients ages 18 months through 72 months who are at risk for developmental delay based on concerns of a parent, caregiver, or healthcare provider.
The device is not intended for use as a stand-alone diagnostic device but as an adjunct to the diagnostic process. The device is for prescription use only (Rx only).
Contraindications
There are no contraindications to using Canvas Dx.
Precautions, Warnings
The Device is intended for use by healthcare professionals trained and qualified to interpret the results of a behavioral assessment examination and to diagnose ASD.
The Device is intended for use in conjunction with patient history, clinical observations, and other clinical evidence the HCP determines are necessary before making clinical decisions. For instance, additional standardized testing may be sought to confirm the Device output, especially when the Device result is not Positive or Negative for ASD.
Canvas Dx is intended for patients with caregivers who have functional English capability (8th grade reading level or above) and have access to a compatible smartphone with an internet connection in the home environment.
The Device may give unreliable results if used in patients with other conditions that would have excluded them from the clinical study.
Among those conditions are the following:
• Suspected auditory or visual hallucinations or with prior diagnosis of childhood onset schizophrenia
• Known deafness or blindness
• Known physical impairment affecting their ability to use their hands
• Major dysmorphic features or prenatal exposure to teratogens such as fetal alcohol syndrome
• History or diagnosis of genetic conditions (such as Rett syndrome or Fragile X)
• Microcephaly
• History or prior diagnosis of epilepsy or seizures
• History of or suspected neglect
• History of brain defect injury or insult requiring interventions such as surgery or chronic medication
The Device evaluation should be completed within 60 days of the time it is prescribed because neurodevelopmental milestones change rapidly in the indicated age group.
Screenshots
Canvas Dx Häufige Fragen
-
Ist Canvas Dx kostenlos?
Ja, Canvas Dx ist komplett kostenlos und enthält keine In-App-Käufe oder Abonnements.
-
Ist Canvas Dx seriös?
Nicht genügend Bewertungen, um eine zuverlässige Einschätzung vorzunehmen. Die App benötigt mehr Nutzerfeedback.
Danke für die Stimme -
Wie viel kostet Canvas Dx?
Canvas Dx ist kostenlos.
-
Wie hoch ist der Umsatz von Canvas Dx?
Um geschätzte Einnahmen der Canvas Dx-App und weitere AppStore-Einblicke zu erhalten, können Sie sich bei der AppTail Mobile Analytics Platform anmelden.

Benutzerbewertung
Die App ist in Rumänien noch nicht bewertet.

Bewertungsverlauf
Canvas Dx Bewertungen
Keine Bewertungen in Rumänien
Die App hat noch keine Bewertungen in Rumänien.
Store-Rankings

Ranking-Verlauf
App-Ranking-Verlauf noch nicht verfügbar

Kategorien-Rankings
App ist noch nicht gerankt
Canvas Dx Installationen
Letzte 30 TageCanvas Dx Umsatz
Letzte 30 TageCanvas Dx Einnahmen und Downloads
Gewinnen Sie wertvolle Einblicke in die Leistung von Canvas Dx mit unserer Analytik.
Melden Sie sich jetzt an, um Zugriff auf Downloads, Einnahmen und mehr zu erhalten.
Melden Sie sich jetzt an, um Zugriff auf Downloads, Einnahmen und mehr zu erhalten.
App-Informationen
- Kategorie
- Medical
- Herausgeber
- Cognoa
- Sprachen
- English, Spanish
- Letzte Veröffentlichung
- 2.1.0 (vor 2 Monaten )
- Veröffentlicht am
- Apr 29, 2024 (vor 1 Jahr )
- Auch verfügbar in
- Vereinigte Staaten, Australien, Belgien, Kanada, Schweiz, Tschechien, Dänemark, Ägypten, Spanien, Vereinigtes Königreich, Griechenland, Ungarn, Irland, Polen, Portugal, Rumänien, Russland, Schweden, Ukraine, Italien, Deutschland
- Zuletzt aktualisiert
- vor 2 Monaten
This page includes copyrighted content from third parties, shared solely for commentary and research in accordance with fair use under applicable copyright laws. All trademarks, including product, service, and company names or logos, remain the property of their respective owners. Their use here falls under nominative fair use as outlined by trademark laws and does not suggest any affiliation with or endorsement by the trademark holders.
- © 2025 AppTail.
- Unterstützung
- Privacy
- Terms
- All Apps